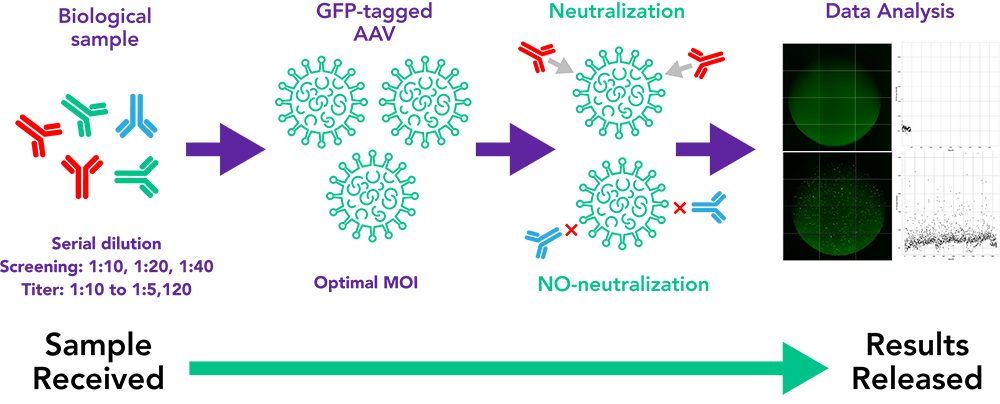
VRL offers a cell-based in vitro assay in two formats for the detection of anti-AAV neutralizing antibodies:
- The screening assay (qualitative neutralization assay): the result is reported as positive or negative for the 3 dilutions (1:10, 1:20, 1:40) of each serum sample.
- The neutralizing antibody titer (or 50% inhibitory dose [ID50]) assay: (quantitative neutralization assay): each serum sample is 2-fold serial diluted from 1:10-1:5120, the result is reported as a titer value at which 50% inhibition of virus transduction is calculated.
VRL is one of the FIRST Commercial Laboratories Offering AAV Neutralizing Antibody Testing
What does that mean for you? It means that you can take advantage of the expertise, knowledge and efficiencies of a commercial laboratory. Including:
Fast Turnaround Time
Get your results in days, not weeks. When time is of the essence, count on VRL to come through every time.
Competitive Pricing
We have the equipment and expertise necessary to efficiently process results. This saves us time – and it saves you money.
Ready & Available
We value our customer relationships, and that means being available when you need us most.
And Available for GLP Studies!

VRL Cell-based NAb assay for AAV’s